Nucleus of Hydrogen is an exception because it contains only one proton and no neutron. The mass of neutron and proton is almost the same. These neutral particles are called neutrons. In 1932 Chadwick proved the existence of neutral particles in the nucleus. Thus it was considered that the nucleus consists of z number of positively charged particles. Where e is the magnitude of the charge on one electron. Rutherford’s model of an atom is also called as a planetary model of an atom.įrom the x-ray technique, it is observed that the charge on the nucleus is Ze and it is positive. 
The total positive charge of the nucleus is equal to the total negative charge on orbiting electrons. The whole mass of an atom is concentrated in the nucleus. Around the nucleus, there is empty space in which the negatively charged electrons revolve in different orbits. The atom consists of a centrally located positively charged nucleus. This concludes that the nucleus is very small as compared to the volume of the atom.įrom the observations of the above experiment, Rutherford put forward the concept of his atomic model. Very few alpha particles were rebounded i.e. This massive +ve charge is at the centre of the atom and called the nucleus. This larger deflection is possible only if alpha-particles collide with heavy and positively charged particles inside the atom because like charges only repel each other. The scattering of alpha particles takes place. The scattered alpha particles were detected by a detector fixed on a stand. The deviation of alpha particles from their original path is called the scattering angle. They observed that most of the alpha particles just passed through without any deviation as if there is empty space. A few alpha particles were deflected through smaller angles. A few alpha particles deviated through larger angles. Scientific facts such as the atomic structure, ionization energy, conductivity and thermal properties are also included.A narrow beam of alpha particles from the radioactive source was incident on a thin gold foil. Francium information, including technical data, properties, research, and other useful facts are discussed below. Due to its rarity and instability, commercial applications for francium do not exist. Out of all of the naturally occurring elements, francium is the most unstable. 
Outside of the laboratory, only trace amounts of francium can be found in uranium and thorium ores. Francium has the lowest electronegativitiy of all known elements and it was the last element discovered in nature. Francium was discovered by Marguerite Perey of the Curie Institute in Paris, France in 1939 although its existence was predicted by Mendeleev during the 1870's.įrancium, formerly known as eka-caesium and actinium K, is the second rarest naturally occurring element, the first being Astatine. 
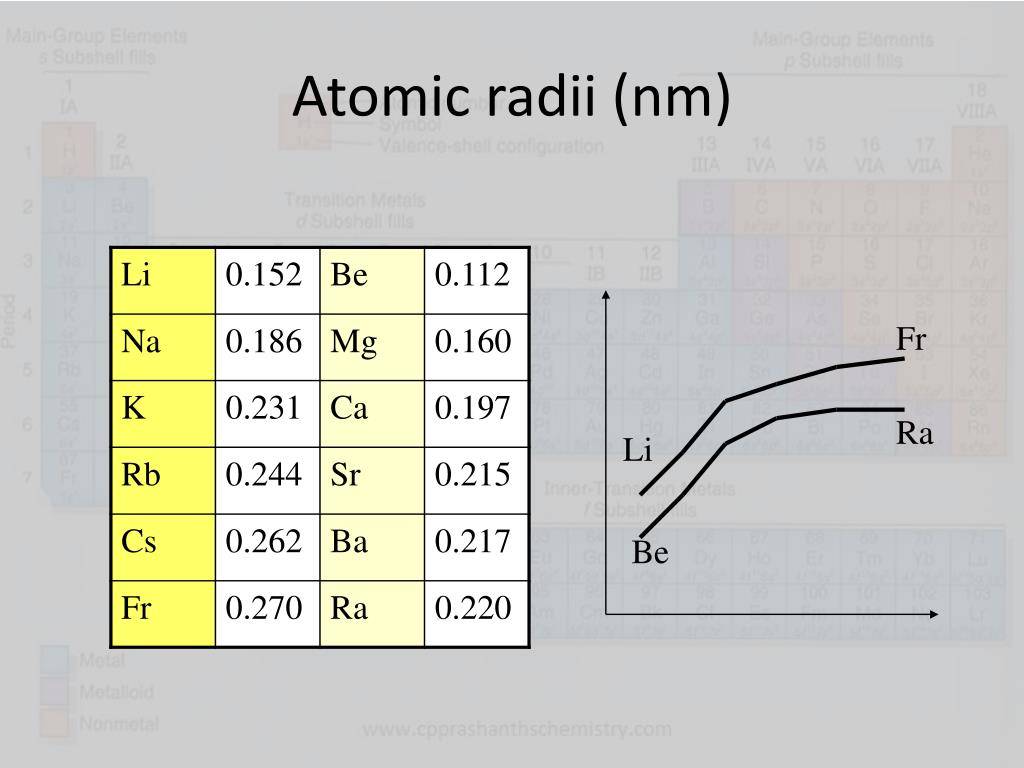
The francium atom has a radius of 270.pm and it's Van der Waals radius is 348.pm. In its elemental form francium's CAS number is. The number of electrons in each of Francium's shells is 2,8,18,32,18,8,1 and its electronic configuration is 1s 2 2s 2p 6 3s 2p 6d 10 4s 2p 6d 10f 14 5s 2p 6d 10 6s 2p 6 7s 1. The element was discovered by French chemist Marguerite Catherine Perey of the Curie Institute in 1939 while analyzing the decay of actinium named for her country of origin, Francium would become the last naturally-occurring element to be discovered rather than being synthesized in a laboratory.įrancium is a S-Block, Group 1, Period 7 element. There are no commercial applications for francium because of its instability.ĭmitri Mendeleev predicted the existence of Francium in the 1870's, calling it “eka-caesium” because of its similarity to cesium (“eka” meaning one element down on the periodic table). Thus, scientists estimate that francium’s theoretical properties are similar to the other members of the alkali metals family. The element has never been observed in bulk with the lowest electronegativity of any element on the periodic table, it would immediately vaporize due to the extreme heat of decay. As such, only 20-30 grams of the element are present at any given time in the earth’s crust, limited to trace amounts in uranium and thorium ores.įrancium can be produced in the laboratory via bombardment of thorium with protons or radium with neutrons, but only in small quantities-the largest amount ever synthesized was a cluster of 30,000 atoms. A product of a disintegration of actinium, which undergoes beta-disintegration 99% of the time, francium is so highly radioactive that its most stable isotope (Francium-233, formerly known as actinium-K) has a half-life of merely 22 minutes. American Elements: The Materials Science Company™ | Certified bulk & lab quantity manufacturer of metals, chemicals, nanoparticles & other advanced materialsįrancium is the second rarest and the most unstable naturally occurring element on the periodic table only astatine is rarer. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
